Термодинамические константы некоторых веществ
Вещество
Al
28,31
Al2 O3
–1675,00
71,10
–1576,40
Al2 (SO4 )3
–3434,00
239,20
–3091,90
BaCO3
–1210,85
112,13
–1132,77
BaO
–553,54
70,29
–525,84
BeO
–598,73
14,14
–596,54
C (графит)
5,74
С (алмаз)
1,83
2,37
2,83
CO
–110,53
197,55
–137,15
CO2
–393,51
213,66
–394,37
CS2 (г)
116,70
237,77
66,55
CS2 (ж)
88,70
151.04
64,61
CH4
–74,85
186,27
–50,85
C2 H2
226,75
200,82
209,21
C2 H4
52,30
219,42
68,14
C2 H6
–84,67
229,49
–32,93
C6 H6
49,03
173,26
124,38
CH3 OH
–201,00
239,76
–162,38
CH3 COH
–115,90
218,78
–109,94
C2 H5 OH
–234,80
281,38
–167,96
C4 H9 OH
–274,43
363,17
–157,73
C6 H12 O6
–1273,00
–
–919,5
C12 H22 O11
–2220,87
359,82
–1529,67
C6 H5 NH2
31,09
192,29
149,08
СaO
–635,10
39,70
–604,20
Ca(OH)2
–986,20
83,40
–896,76
CaC2
–62,70
70,30
–67,80
CaCO3
–1206,83
91,71
–1128,35
СdО
–258,99
54,81
–229,33
Cl2
222,98
Cr
23,64
Cr2 O3
–1140,56
82,17
–1058,97
Cu
33,30
CuO
–165,30
42,64
–127,19
Fe
27,15
FeO
–263,68
58,79
–244,35
Fe2 O3
–822,16
87,45
–740,34
Fe3 O4
–1117,13
146,19
–1014,17
FeS
–100,42
60,29
–100,78
H2
130,52
HCl (г)
–92,31
186,79
–95,30
HCl (ж)
–167,50
55,20
–132,10
H2 O (г)
–241,83
69,94
–237,19
Окончание приложения 2
Вещество
H2 O (ж)
–285,84
188,72
–228,59
H2 S (г)
–20,15
205,64
–33,02
H2 S (ж)
–39,33
122,20
–27,36
Hg
76,10
Hg2 Cl2
–264,85
185,81
–210,66
HgCl2
–230,12
144,35
–185,17
MgO
–601,49
27,07
–569,27
MgCO3
–1095,85
65,10
–1012,15
N2
191,50
N2 O
82,01
219,83
104,12
NO
91,21
210,64
87,58
NO2
33,50
240,20
51,20
NH3
–45,94
192,66
–16,48
NH4 Cl
–314,22
95,81
–203,22
NH4 NO3
–365,43
151,04
–183,93
(NH4 )2 CO3
–314,22
95,81
–203,22
Na2 O
–430,60
71,10
–376,60
Na2 CO3
–1129,00
136,00
–1047,70
O2
205,04
PCl3
–277,00
311,70
–286,27
PCl5
–369,45
362,90
–324,55
PbO
–217,61
68,70
–188,20
PbS
–100,42
91,21
–98,77
S
31,88
SO2
–296,90
248,07
–300,21
SO3
–395,85
256,69
–371,17
TiO2
–944,80
50,33
–889,49
Приложение 3
Дата публикования: 2015-10-09 ; Прочитано: 482 | Нарушение авторского права страницы
studopedia.org - Студопедия.Орг - 2014-2024 год. Студопедия не является автором материалов, которые размещены. Но предоставляет возможность бесплатного использования
(0.005 с) ...

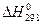 ,
кДж/моль
,
кДж/моль
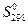 ,
Дж/(моль·K)
,
Дж/(моль·K)
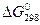 ,
кДж/моль
,
кДж/моль
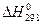 ,
кДж/моль
,
кДж/моль
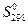 ,
Дж/(моль·K)
,
Дж/(моль·K)
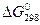 ,
кДж/моль
,
кДж/моль